Electronic Handedness in Copper-Silver Combo
The electrical properties of a thin layer of copper compound on a silver surface are exposed in high resolution scans. The images reveal surprising differences between the two samples. Although superficially similar, the two patterns are mirror images, much as right and left hands mirror each other.
This “handedness,” which is also known as chirality, is surprising because the copper compound and the underlying silver surface are built of molecules that lack chirality. Previously, scientists had assumed that molecules would have to have a chiral structure in order to produce handed patterns on surfaces. The research that produced these images shows that chirality can sometimes be a purely electronic effect.
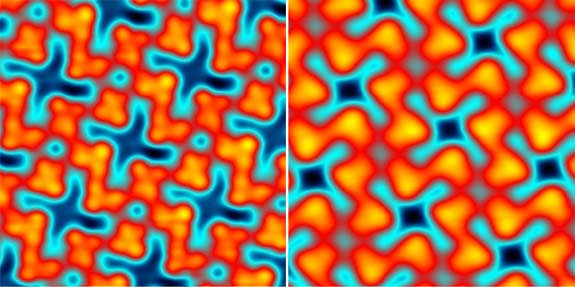
Image Credit: A. Mugarza, C. Krull, S. Stepanow, G. Ceballos, and P. Gambardella, CIN2
These images show two types of molecular assembly configurations. Each reaction was observed after a copper phthalocyanine (CuPc) monolayer was deposited on a silver (Ag(100)) surface.
This research is supported by the Spanish Ministerio de Ciencia e Innovacio´n (FIS2006-12117- C04-01 and MAT2007-62341) and the European Research Council (StG 203239).
